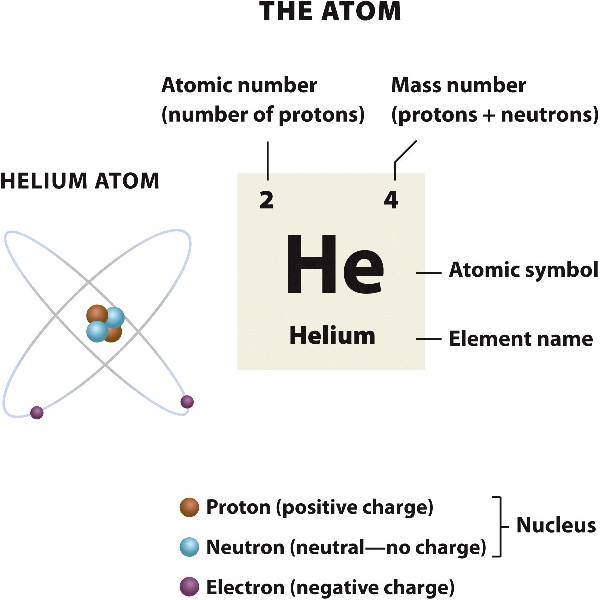
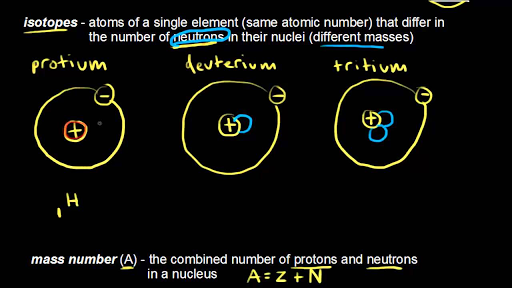
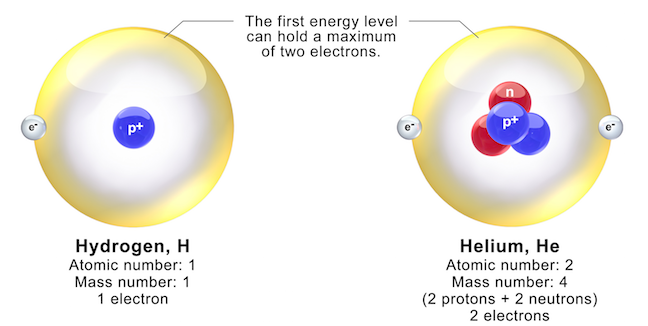
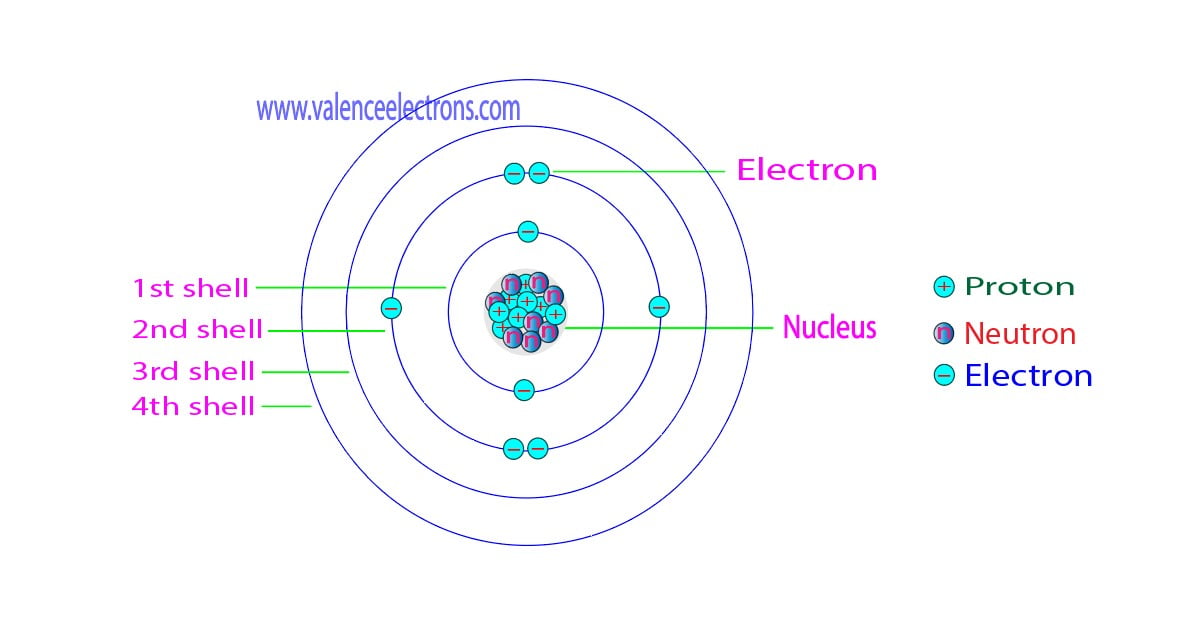
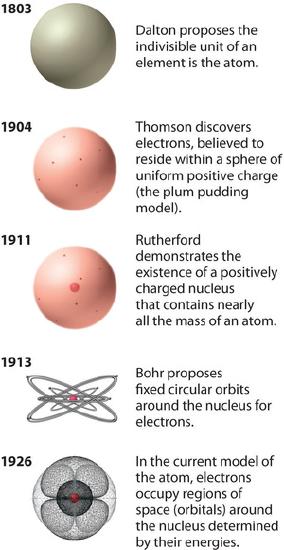
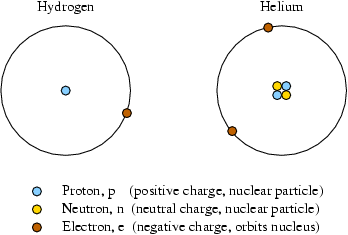
Isotopes Chapter 4, Section 3 Pg Review of Atoms Atoms are made of protons, neutrons, and electrons. – Protons are +, electrons are –, and. - ppt download

Displaying Isotopic Abundance Percentages with Bar Charts and Pie Charts | The Chemical Statistician