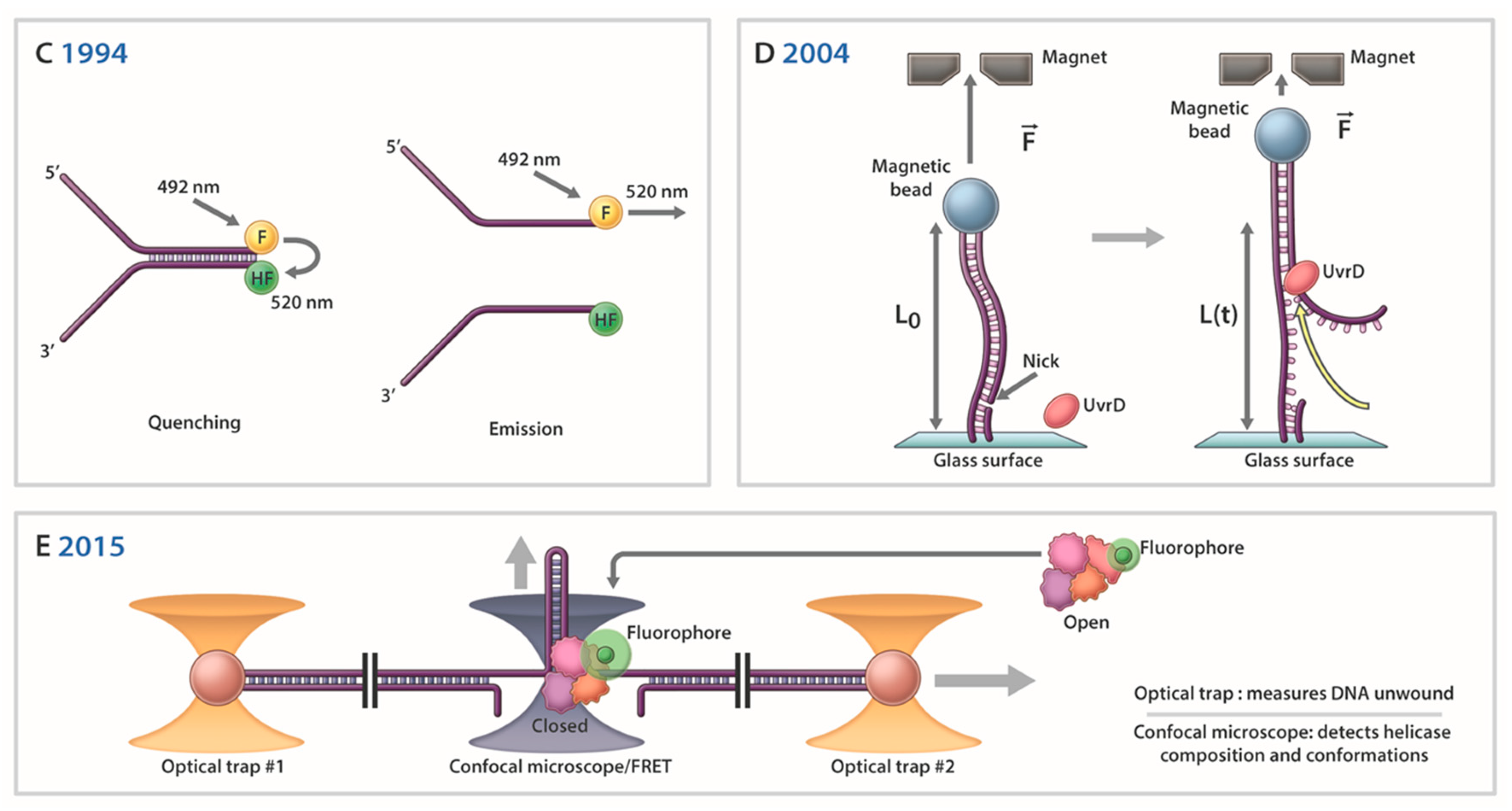
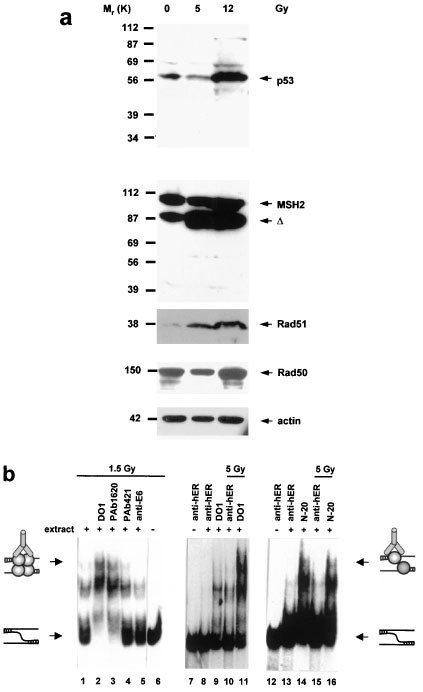
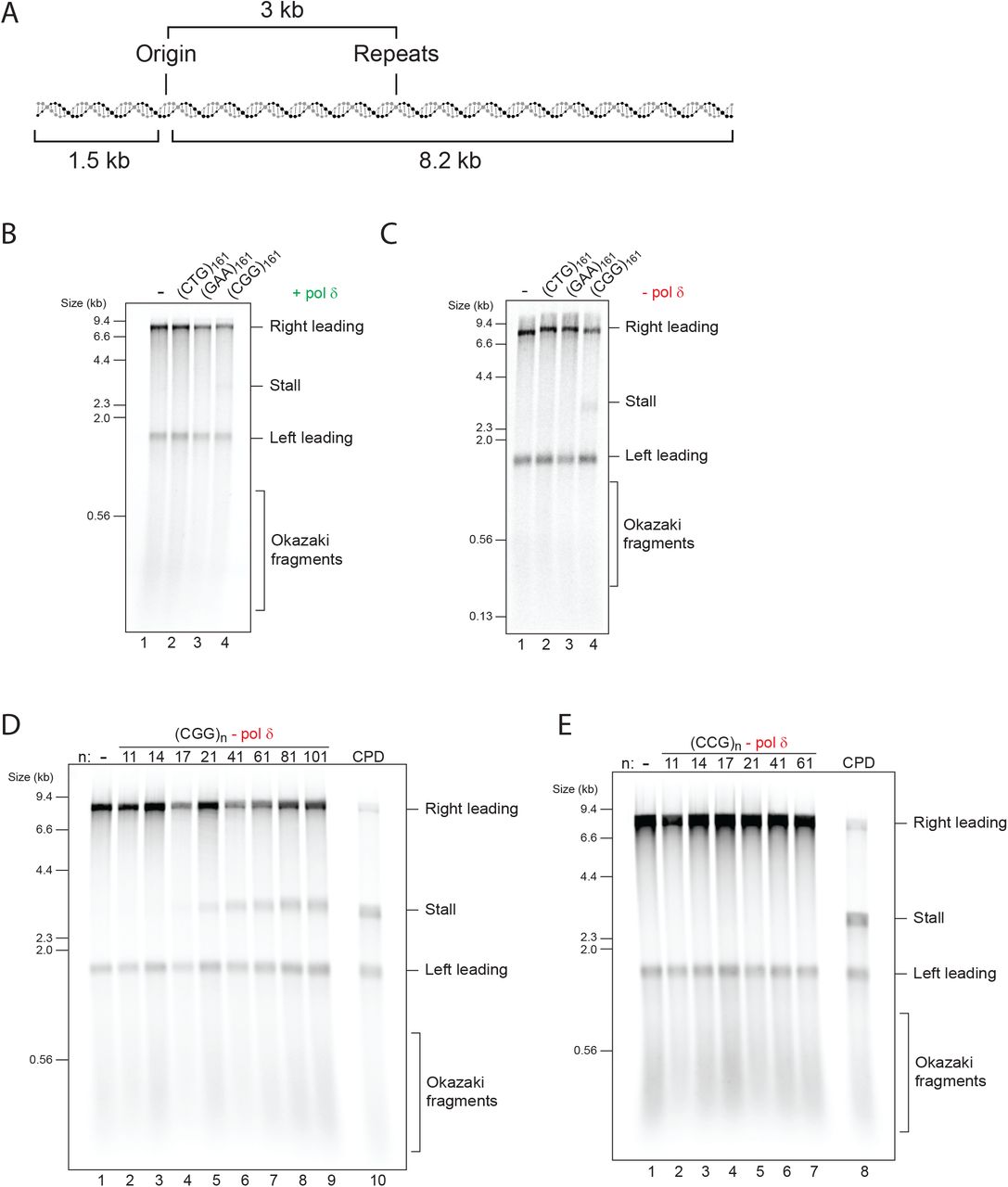
Xeroderma Pigmentosum Complementation Group A Protein (XPA) Modulates RPA-DNA Interactions via Enhanced Complex Stability and Inhibition of Strand Separation Activity* - Journal of Biological Chemistry
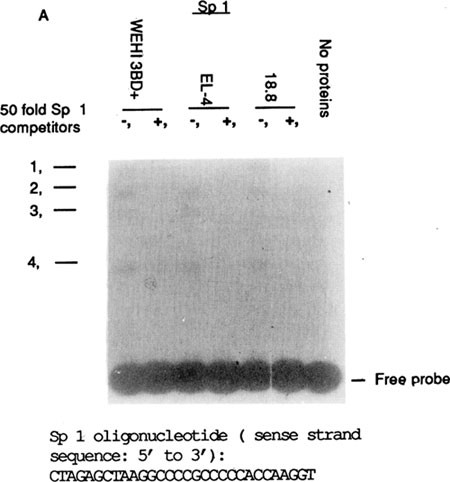
FT I, anovel positive myeloid-lineage-specific transcription regulatory element within the mouse myeloperoxidase gene enhancer, En 1 | Cell Research

Insights into the Degradation of Amorphous Silicon Passivation Layer for Heterojunction Solar Cells - Bernardini - 2019 - physica status solidi (a) - Wiley Online Library

The RepE Initiator Is a Double-stranded and Single-stranded DNA-binding Protein That Forms an Atypical Open Complex at the Onset of Replication of Plasmid pAMβ1 from Gram-positive Bacteria - ScienceDirect

The RepE Initiator Is a Double-stranded and Single-stranded DNA-binding Protein That Forms an Atypical Open Complex at the Onset of Replication of Plasmid pAMβ1 from Gram-positive Bacteria - ScienceDirect

The C-terminal Extension (CTE) of the Nuclear Hormone Receptor DNA Binding Domain Determines Interactions and Functional Response to the HMGB-1/-2 Co-regulatory Proteins - ScienceDirect
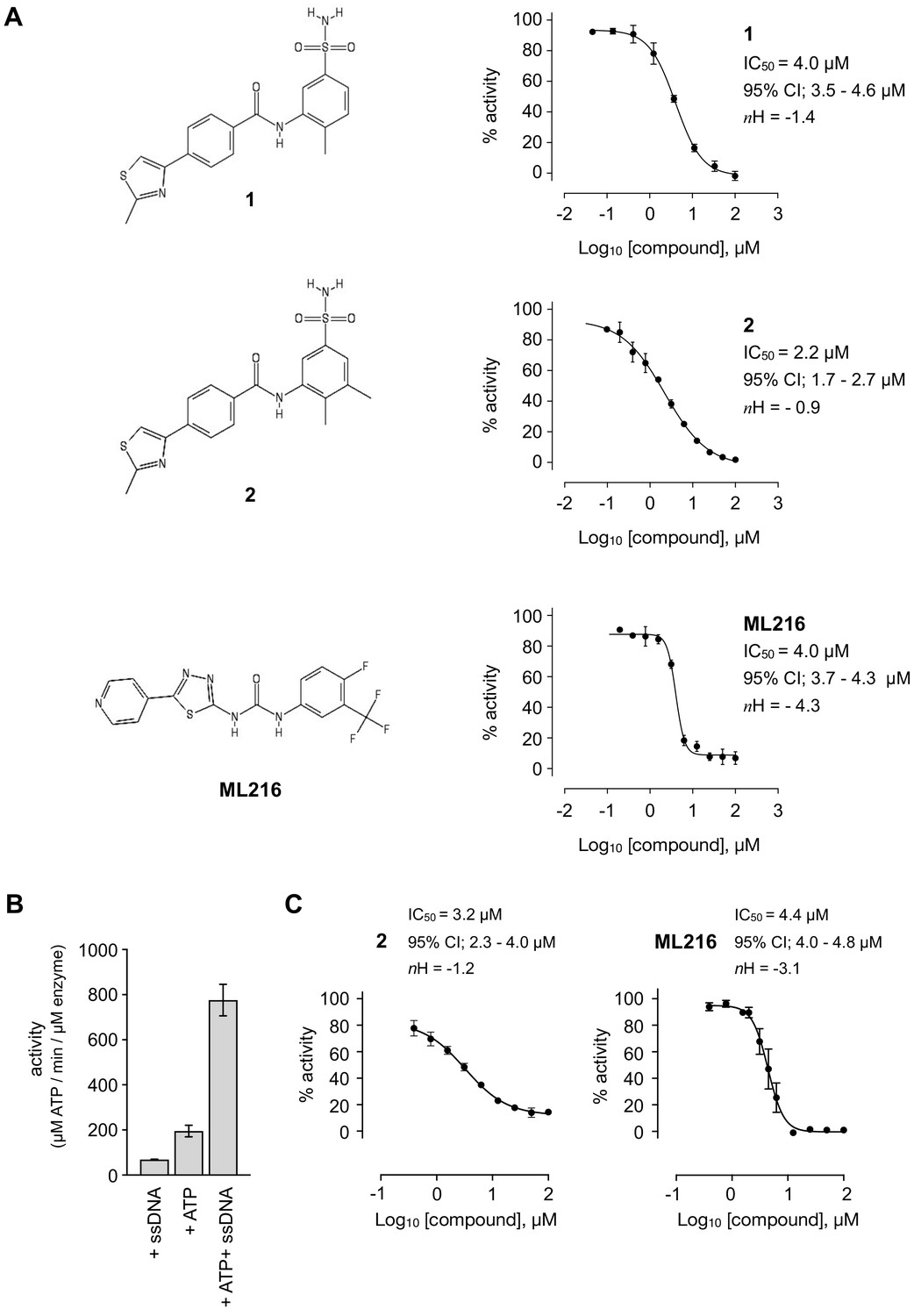
Uncovering an allosteric mode of action for a selective inhibitor of human Bloom syndrome protein | eLife
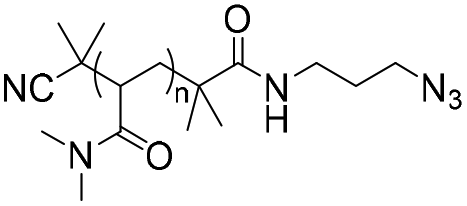
Synthetic approaches for copolymers containing nucleic acids and analogues: challenges and opportunities - Polymer Chemistry (RSC Publishing) DOI:10.1039/D0PY01707H

The RepE Initiator Is a Double-stranded and Single-stranded DNA-binding Protein That Forms an Atypical Open Complex at the Onset of Replication of Plasmid pAMβ1 from Gram-positive Bacteria - ScienceDirect

PDF) Liu W, Wang G, Yakovlev AGIdentification and functional analysis of the rat caspase-3 gene promoter. J Biol Chem 277:8273-8278
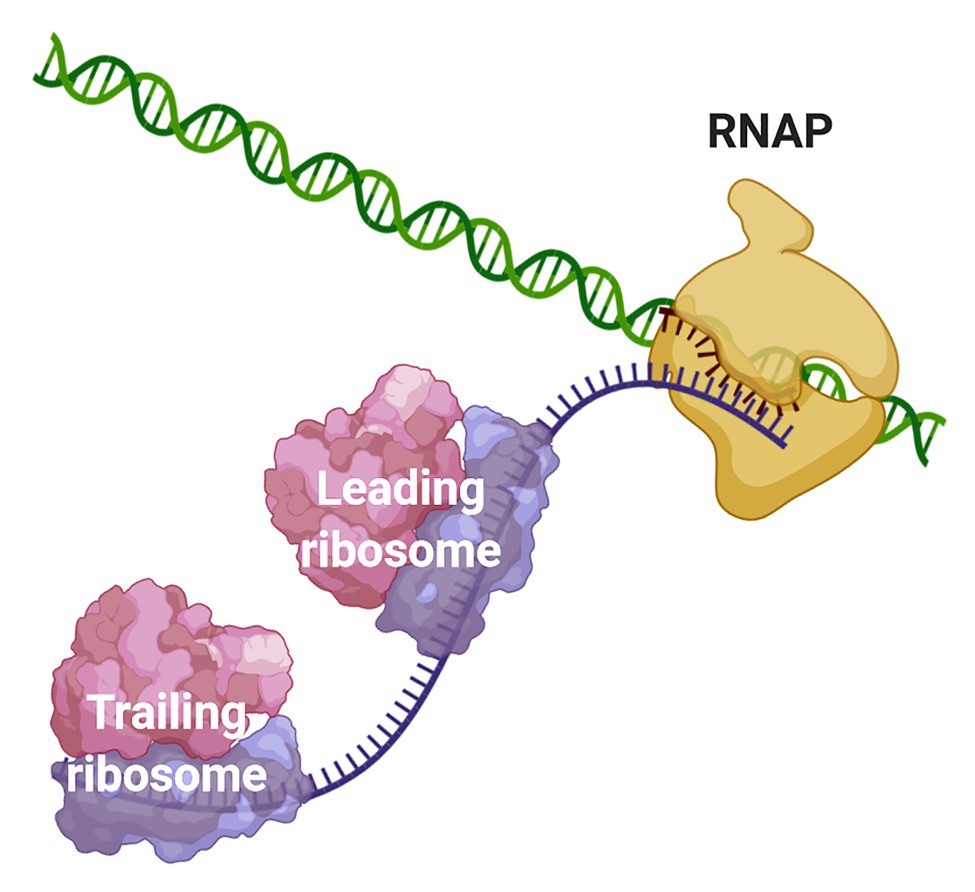
Frontiers | Coupled Transcription-Translation in Prokaryotes: An Old Couple With New Surprises | Microbiology

Elimination of nonspecific bands in non-radioactive electrophoretic mobility shift assays using the digoxigenin system. - Abstract - Europe PMC

Interaction of HelQ helicase with RPA modulates RPA-DNA binding and stimulates HelQ to unwind DNA through a protein roadblock | bioRxiv

Phosphorylation of the Transcription Factor NFATp Inhibits Its DNA Binding Activity in Cyclosporin A-treated Human B and T Cells - ScienceDirect

Action of site‐specific recombinases XerC and XerD on tethered Holliday junctions | The EMBO Journal
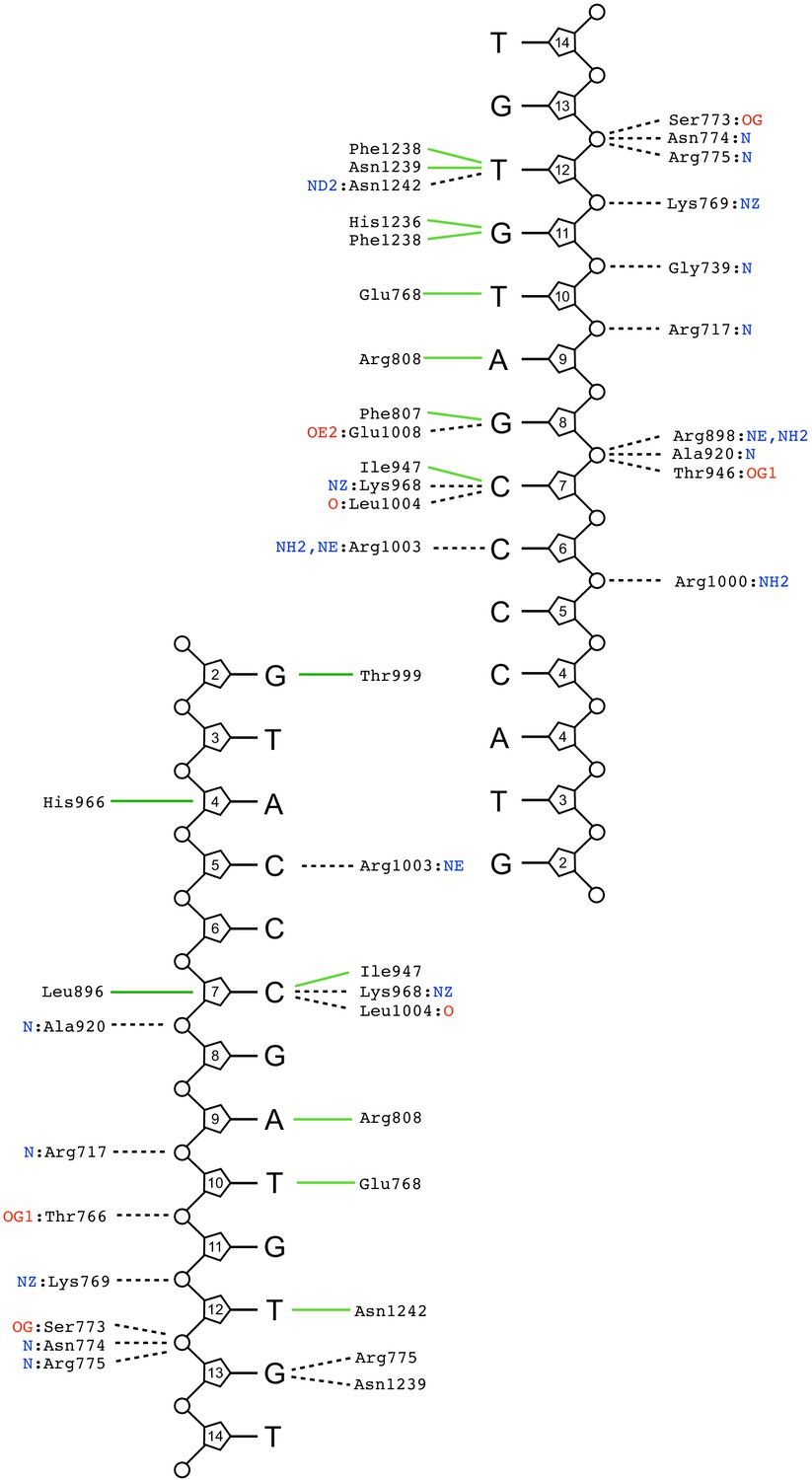
Uncovering an allosteric mode of action for a selective inhibitor of human Bloom syndrome protein | eLife

Drosophila Octamer Elements and Pdm-1 Dictate the Coordinated Transcription of Core Histone Genes - ScienceDirect
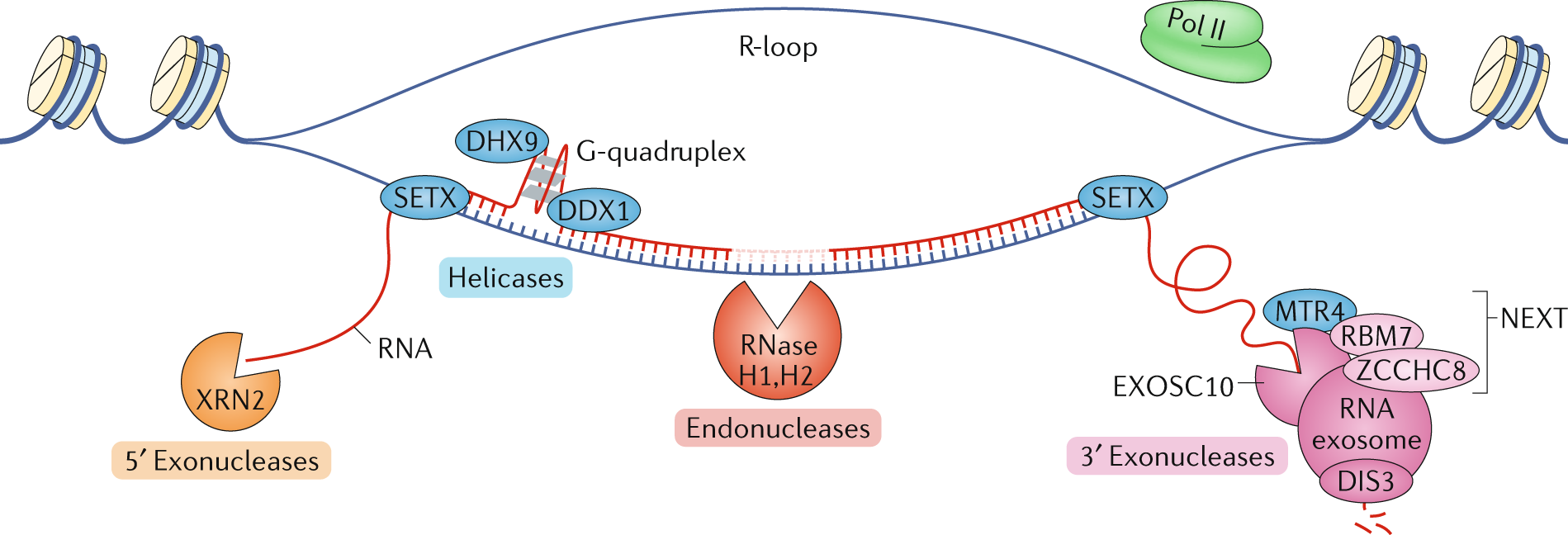
Regulation of long non-coding RNAs and genome dynamics by the RNA surveillance machinery | Nature Reviews Molecular Cell Biology